Series of outstanding presentations from PCORI's electronic data workshop.
Monday, July 23, 2012
Thursday, June 7, 2012
Query Health at the HITPC / HITSC Clinical Quality Hearing
From testimony at the Health IT Policy Committee and Health IT Standards Committee Clinical Quality Hearing, June 7, 2012.
Query Health is working to establish standards to "send questions to the data" while keeping patient level information safe at the data source. Distributed query networks are using these standards in pilots for insights on diabetes and hypertension, national and regional situation awareness, post-market surveillance and dynamic querying for quality measures.
How can the measure development process be improved?
The policy and standards committees have the opportunity to introduce strategic changes that result in agile, responsive, clinically relevant measures in Stage 3.
The clinical quality measure development process today is slow and unresponsive to the rapidly evolving state of medicine in this country. Measures may take one to two years to define, and once defined, measures then take several more years to move through the regulatory cycle, be incorporated into EHR systems, be deployed to providers and then finally implemented for reporting.
Quality measures, even in their latest most formal expression using the Health Quality Measure Format (HQMF), are impossible for a system to digest “automagically”, as HQMF is verbose and not fully computable, with aspects of the measure even described in text. Ambiguity in measure specification leads to multiple interpretations by providers and thus variability, which then requires rework during implementation of the measure in the field.
EHR developers who work with quality measures have described the need for greater clarity and specificity on the supporting data requirements up front, and validation that required data elements can be effectively collected in the provider workflow.
Measure development can also be improved by focusing on a common set of building blocks which could be used to create simple computable queries, which could in turn serve as the foundation for more complex queries. This will also help us to mature the queries without having to re-implement and redefine every concept as part of each individual complex query.
How can measures better leverage electronic health record capabilities?
In collaboration with HL7, NQF and CMS, Query Health standards will enable Health IT vendors to dynamically respond to queries, including queries that align with quality measures. So assuming the data is being captured, the quality measure cycle time could go from years to truly a matter of days. The ability to generate measures nationally in a short cycle time has powerful benefits for patients and patient populations while enabling researchers and healthcare organizations to substantially reduce costs and increase speed.
Blackford talked about the importance of having an externalized set of target data that could deal with the curly braces problem. Query Health standards do just that in a manner that is aligned with the Quality Data Model and Consolidated CDA. Query Health standards provide a road map to better leverage EHR capabilities for dynamic querying of the EHR for quality measures. The standards include the questions (a “new” more parsimonious HQMF), the target data (ONC’s Clinical Element Data Dictionary or CEDD), the results (QRDA Categories 2 & 3) and the Query Envelope.
A Query Health pilot being conducted by Allscripts will evaluate Query Health standards and target data to deliver sample quality measures.
How can the measurement infrastructure and data be leveraged for other types of improvement?
Quality measures are an important class of aggregate measures that can be immensely valuable. Clinical quality measure queries, with the Query Health standards applied, align with the Stage 3 goals for improved outcomes and establishing a learning health system through rapid feedback mechanisms.
Pooled “big data” in healthcare has its benefits but also has several drawbacks. “Big data” is typically managed in large pooled data sets, combining data from many settings of care. While there are terrific applications for pooled data, including registries and other successful use of large research and commercial databases, there are also critical issues of policy and strategy that must be resolved. Query Health standards can serve as the safe on-ramp to “big data”.
Ultimately, we're at a defining moment for standards that will enable quality measures and big data analytics in a distributed environment. Researchers will be able to leverage these standards to “send questions to the data.” Questions can be sent to numerous data sources including EHRs, HIEs, PHRs, payers’ clinical record or any other clinical record. Aggregate responses leave patient level information secure behind the data source’s firewall. Those responses can support questions related to disease outbreak, quality, research, post-market surveillance, performance, utilization, public health, prevention, resource optimization and many others. The opportunities are truly endless. Thank you.
Labels:
#QueryHealth
Thursday, May 31, 2012
Query Health - by the numbers
I'm finishing up a year with the Office of the National Coordinator for Health IT. Thought you'd be interested in this blog post, summing up the work on Query Health.
Query Health – by the
numbers
From Rich Elmore, Coordinator, Query Health
I wanted to share with you an update on Query Health - - by
the numbers.
|
One
|
Transformative
concept
|
Sending questions to the data
|
|
Two
|
Operational
documents
|
1) Data
use agreement
2) Operational
guidance
|
|
Three
|
Reference
Implementations
|
1) i2b2
2) PopMedNet
3) hQuery
|
|
Four
|
Standards
|
1) Question
(HQMF)
2) Data
(CEDD)
3) Results
(QRDA Cat 2&3)
4) Query
Envelope
|
|
Five
|
Pilots
|
1) NYC/NYS
DPH
2) Mini-Sentinel
(FDA/BID)
3) CDC
BioSense 2
4) Mass
DPH
5) CQM
(Allscripts)
|
The Query Health technical work group is making fantastic
progress on the reference implementations. We’ve also had recent exciting
news with ONC and HL7 working jointly on preparing HQMF and QRDA for ballot,
and ONC and NQF working jointly on aligning CEDD with QDM.
The Query Health pilots are poised to ask and answer
important questions related to diabetes, hypertension, post-market
surveillance, situation awareness and clinical quality measures.
Most importantly, Query Health aligns with the concept of a
learning health system, focusing on improving patient and population outcomes.
All of this is the result of the energized, expert, engaged
community that have provided shape and direction for distributed population
queries. Thank you to all of the Query Health members and support team
for your outstanding contributions!
The time I had promised to ONC has gone by so quickly.
I am thrilled that Feik (John Feikema) is taking the reins. We’ve been
working together on the transition for the past month and the project will not
miss a beat.
Onward!
Rich
Labels:
#QueryHealth
Sunday, March 11, 2012
Todd Park, Chief Technology Officer for the U.S.
Todd Park recently assumed the role of Chief Technology Officer (CTO) for the United States reporting to the President. Last April, Todd (then CTO of Health and Human Services), Josh Seidman and I participated on a panel moderated by Sean Nolan on Citizen-Centric Health: How Public/Private Partnerships are Changing the Game. Todd's energy and passion on that panel were so great, that it is worthwhile reprising here.
Monday, March 5, 2012
Standards for Distributed Population Health Queries
Originally published in Faster Cures' "bloggersation" regarding: What is the most important thing that could happen in 2012 to ensure better utilization of big data—housed in EMRs or other platforms—for drug development?
“Big data” is typically managed in large pooled data sets, combining data from many settings of care. While there are terrific applications of pooled data, including registries and successful use of large research databases, there are critical issues of policy and strategy. Pooled “Big data” in healthcare has its benefits but also has several drawbacks.
From a policy perspective, pooled data approaches are problematic. Large pools of PHI are targets for attack from bad actors. Also, many PHI-holders have their own consent agreements with their patients. It is difficult to manage these different consent agreements when pooling PHI in one place. Additionally, HIPAA requires covered entities to control the flow of PHI, either directly or through agreements. When data is pooled, the party pooling the data must have a business associate agreement or data use agreement (in the case of research databases) with each covered entity that contributes data to the pool, with the same (or similar terms). This can be impracticable for the third party or undesirable for covered entities, as they often have to agree to non-negotiable terms in the agreement in order to pool their data.
From a strategic standpoint, pooled data is inflexible, stale and inaccurate. Pooled data approaches aren’t generally sustainable: the benefits of pooled approaches are too indirect to support the operational costs and complexity. Furthermore, health care organizations are unwilling to lose control of their information not just for policy reasons, but also due to competitive considerations.
But the absence of a standards-based alternative has given rise to pooled data approaches with all of these substantial drawbacks.
2012 is the defining moment for new standards that will enable big data analytics in a distributed environment. An ONC sponsored open government initiative, Query Health, is defining the standards and specifications for distributed population queries. Researchers will be able to leverage these standards to be “send questions to the data”. Questions can be sent to data sources including EHRs, HIEs, PHRs, payers’ clinical record or any other clinical record. Aggregate responses leave patient level information secure behind the data source’s firewall. Aggregate responses support questions related to disease outbreak, quality, CER, post-market surveillance, performance, utilization, public health, prevention, resource optimization and many others.
The path for these new standards will dramatically cut cycle time for deployment of new questions from years to days – making possible support for a learning health system.
The focus of 2012 should be laying the foundation for success: defining the standards and services for distributed population health queries. This is one extremely impactful way to leverage the potential of big data for research. For more information, visit QueryHealth.org.
“Big data” is typically managed in large pooled data sets, combining data from many settings of care. While there are terrific applications of pooled data, including registries and successful use of large research databases, there are critical issues of policy and strategy. Pooled “Big data” in healthcare has its benefits but also has several drawbacks.
From a policy perspective, pooled data approaches are problematic. Large pools of PHI are targets for attack from bad actors. Also, many PHI-holders have their own consent agreements with their patients. It is difficult to manage these different consent agreements when pooling PHI in one place. Additionally, HIPAA requires covered entities to control the flow of PHI, either directly or through agreements. When data is pooled, the party pooling the data must have a business associate agreement or data use agreement (in the case of research databases) with each covered entity that contributes data to the pool, with the same (or similar terms). This can be impracticable for the third party or undesirable for covered entities, as they often have to agree to non-negotiable terms in the agreement in order to pool their data.
From a strategic standpoint, pooled data is inflexible, stale and inaccurate. Pooled data approaches aren’t generally sustainable: the benefits of pooled approaches are too indirect to support the operational costs and complexity. Furthermore, health care organizations are unwilling to lose control of their information not just for policy reasons, but also due to competitive considerations.
But the absence of a standards-based alternative has given rise to pooled data approaches with all of these substantial drawbacks.
2012 is the defining moment for new standards that will enable big data analytics in a distributed environment. An ONC sponsored open government initiative, Query Health, is defining the standards and specifications for distributed population queries. Researchers will be able to leverage these standards to be “send questions to the data”. Questions can be sent to data sources including EHRs, HIEs, PHRs, payers’ clinical record or any other clinical record. Aggregate responses leave patient level information secure behind the data source’s firewall. Aggregate responses support questions related to disease outbreak, quality, CER, post-market surveillance, performance, utilization, public health, prevention, resource optimization and many others.
The path for these new standards will dramatically cut cycle time for deployment of new questions from years to days – making possible support for a learning health system.
The focus of 2012 should be laying the foundation for success: defining the standards and services for distributed population health queries. This is one extremely impactful way to leverage the potential of big data for research. For more information, visit QueryHealth.org.
Labels:
Query Health
Saturday, February 25, 2012
Meaningful Use Stage 2
HHS News Release
Health and Human Services Secretary Kathleen Sebelius announced the next steps for providers who are using electronic health record (EHR) technology and receiving incentive payments from Medicare and Medicaid. These proposed rules, from the Centers for Medicaid & Medicaid Services (CMS) and the Office of the National Coordinator for Health Information Technology (ONC), will govern stage 2 of the Medicare and Medicaid Electronic Health Record Incentive Programs.
Health and Human Services Secretary Kathleen Sebelius announced the next steps for providers who are using electronic health record (EHR) technology and receiving incentive payments from Medicare and Medicaid. These proposed rules, from the Centers for Medicaid & Medicaid Services (CMS) and the Office of the National Coordinator for Health Information Technology (ONC), will govern stage 2 of the Medicare and Medicaid Electronic Health Record Incentive Programs.
“We know that broader adoption of electronic health records can save our health care system money, save time for doctors and hospitals, and save lives,” said Secretary Sebelius. “We have seen great success and momentum as we’ve taken the first steps toward adoption of this critical technology. As we move into the next stage, we are encouraging even more providers to participate and support more coordinated, patient-centered care.”
Under the Health Information Technology for Economic and Clinical Health (HITECH) Act, part of the American Recovery and Reinvestment Act of 2009, eligible health care professionals and hospitals can qualify for Medicare and Medicaid incentive payments when they adopt certified EHR technology and use it in a meaningful way. What is considered “meaningful use” is evolving in three stages:
- Stage 1 (which began in 2011 and remains the starting point for all providers): “meaningful use” consists of transferring data to EHRs and being able to share information, including electronic copies and visit summaries for patients.
- Stage 2 (to be implemented in 2014 under the proposed rule): “meaningful use” includes new standards such as online access for patients to their health information, and electronic health information exchange between providers.
- Stage 3 (expected to be implemented in 2016): “meaningful use” includes demonstrating that the quality of health care has been improved.
CMS’ proposed rule specifies the stage 2 criteria that eligible providers must meet in order to qualify for Medicare and/or Medicaid EHR incentive payments. It also specifies Medicare payment adjustments that, beginning in 2015, providers will face if they fail to demonstrate meaningful use of certified EHR technology and fail to meet other program participation requirements. In a November 2011 “We Can’t Wait” announcement (http://www.hhs.gov/news/press/2011pres/11/20111130a.html), the Department outlined plans to provide an additional year for providers who attested to meaningful use in 2011. Under today’s proposed rule, stage 1 has been extended an additional year, allowing providers to attest to stage 2 in 2014, instead of in 2013. The proposed rule announced by ONC identifies standards and criteria for the certification of EHR technology, so eligible professionals and hospitals can be sure that the systems they adopt are capable of performing the required functions to demonstrate either stage of meaningful use that would be in effect starting in 2014.
“Through the Medicare and Medicaid EHR Incentive Programs, we’ve seen incredible progress as over 43,000 providers have received $3.1 billion to help make the transition to electronic health records,” said CMS Acting Administrator Marilyn Tavenner. “There is great momentum as the number of providers adopting this technology grows every month. Today’s announcement will help ensure broad participation and success of the program, as we move toward full adoption of this money-saving and life-saving technology.”
“The proposed rules for stage 2 for meaningful use and updated certification criteria largely reflect the recommendations from the Health IT Policy and Standards Committees, the federal advisory committees that operate through a transparent process with broad public input from all key stakeholders. Their recommendations emphasized the desire to increase health information exchange, increase patient and family engagement, and better align reporting requirements with other HHS programs,” said Farzad Mostashari, MD, ScM, National Coordinator for Health Information Technology. “The proposed rules announced today will continue down the path stage 1 established by focusing on value-added ways in which EHR systems can help providers deliver care which is more coordinated, safer, patient-centered, and efficient.”
The number of hospitals using EHRs has more than doubled in the last two years from 16 to 35 percent between 2009 and 2011. Eighty-five percent of hospitals now report that by 2015 they intend to take advantage of the incentive payments.
A technical fact sheet on CMS’s proposed rule is available at http://www.cms.gov/apps/media/fact_sheets.asp.
A technical fact sheet on ONC’s standards and certification criteria proposed rule is available athttp://www.healthit.gov/policy-research.
The proposed rules announced today may be viewed at www.ofr.gov/inspection.aspx. Comments are due 60 days after publication in the Federal Register.
Wednesday, February 15, 2012
Query Health: Distributed Population Queries
By Michael Buck and Rich Elmore (and originally published in Health Data Management)
Query Health is an Open Government Initiative that is establishing the standards and services for distributed population health queries. Query Health standards will be used to send questions to clinical data sources which return aggregate measures of population health that can be used for many purposes including disease outbreak monitoring, post-market surveillance, comparative effectiveness research, quality and performance measures.
Query Health is pleased to announce the commitment of leading healthcare organizations to pilot the Query Health standards and specifications. Query Health also plans to present its progress on proposed standards and reference implementation at the HIMSS conference in Las Vegas, February 20-24, 2012. And in an unexpected twist, Query Health standards will deliver potential benefits beyond the scope of distributed population queries.
Announcing the first Query Health Pilot
Today, Query Health is announcing that the Primary Care Information Project (PCIP), within the New York City Department of Health and Mental Hygiene, and the New York State Department of Public Health have announced plans to pilot the Query Health standards and reference implementation. PCIP recently won awards for their work on distributed queries including the 2011 HIMSS Public Health Davies Award of Excellence and Healthcare Informatics first place Innovator Award. They will be using Query Health standards to expand their existing population health monitoring network from an existing 1.6 million ambulatory patients to encompass citywide HIE organizational coverage of both inpatient and outpatient encounters. This enhanced system will support optimal allocation of limited public health resources.
What will be shown at HIMSS?
ONC will host a demonstration of Query Health’s progress on the reference implementation including a live demo of distributed query execution.
· ONC Booth Theater on Wednesday February 22nd at 9:45 AM.
· HIMSS Interoperability Showcase Stage on Thursday February 23rd at 9:30 AM
Are there benefits beyond distributed population queries?
The path for a quality measure today is measured in years from the time of measure definition to delivery in vendor systems and deployment in EHRs. Want another quality measure? Wait several years.
In collaboration with HL7, NQF and CMS, Query Health standards will enable Health IT vendors to dynamically respond to queries, including queries that align with quality measures. So assuming the data is being captured, the cycle time could go from years to days.
The ability to generate measures nationally in a short cycle time has powerful benefits for patients, patient populations while enabling researchers and healthcare organizations to substantially reduce costs and increase speed.
What is Query Health?
Query Health was launched September 2011, with approximately 100 committed member organizations representing diverse healthcare stakeholders contributing to the project.
Today, when health researchers develop questions about a population, in many cases they manually pose these questions to care delivery organizations, which employ technical teams to manually generate queries and produce reports. Even where distributed queries are automated, the costs and time to link each data source are unacceptably high. The Query Health Initiative will make this much more efficient: the question can be delivered in a format that will be interpreted automatically by an HIT system. The HIT system will then generate a report with the “answer” to the query.
Questions can be sent to many different types of data sources including providers’ EHRs, payers’ clinical records, personal health records and health information exchanges. Decisions about which queries to process are under control of the data owner and the aggregated results protect patient level data, which remains safely behind data owners’ firewalls.
For more information:
For more information on Query Health initiative, visit the Query Health website http://www.QueryHealth.org or contact admin@siframework.org.
The authors:
Michael Buck is the clinical work group leader for Query Health and director for the NYC/NYS pilot. Dr. Buck is also Biomedical Informatics R&D Manager at New York City Department of Health and Mental Hygiene and Adjunct Associate Research Scientist at Columbia University’s Department of Biomedical Informatics.
Rich Elmore is the ONC Coordinator for Query Health.
Labels:
#QueryHealth,
Query Health
Tuesday, January 10, 2012
Medical Errors Due to Indecipherable Prescriptions
Thanks to e-Patient Dave for the image.
See also the recent report Hospital Incident Reporting Systems Do Not Capture Most Patient Harm which found that "Hospital staff did not report 86 percent of events to incident reporting systems, partly because of staff misperceptions about what constitutes patient harm".
Wednesday, December 14, 2011
The December HIT Standards Committee Meeting
by John Halamka, Life as a Healthcare CIO
We began the meeting by relating our standards trajectory to today's agenda.
Our outstanding standards issues for discussion include
1. Content
Continued discussion of GreenCDA on the wire and overview of Stan Huff's CIMI initiative
Standardizing DICOM image objects for image sharing and investigating other possible approaches (e.g., cloud based JPEG2000 exchange). Consider image transfer standards, image viewing standards, and image reporting standards.
Query Health i.e. I2B2 distributed queries that send questions to data instead of requiring consolidation of data
Simplify the specification for quality measures to enhance consistency of implementation.
The December meeting included an overview of Query Health and Quality measure standards, leaving the discussion of GreenCDA/CIMI and DICOM to our 2012 meetings.
2. Vocabulary
Extend the quality measurement vocabularies to clinical summaries
Lab ordering compendium
The December meeting included a discussion of the lab ordering compendium, leaving the discussion of clinical summary vocabularies to our 2012 meetings.
3. Transport
Specify how the metadata ANPRM be integrated into the health exchange architecture
Additional NwHIN standards development (hearing re Exchange specification complexity, review/oversight of the S&I framework work on Exchange specifications simplification). Further define secure RESTful transport standards.
Accelerate provider directory pilots (Microdata, RESTful query/response that separates the transaction layer from the schema) and rapidly disseminate lessons learned.
The December meeting included an update on the provider directory and certificate components of transport
Our first presentation was an NCVHS update on ACA Section 10109 by Walter Suarez.
The Committee emphasized the need to coordinate NCHVS work and HITSC work given that division between administrative and clinical data is becoming less distinct over time
Our second presentation was an Implementation Workgroup Update by Liz Johnson about testing procedures that support the certification process.
The committee emphasized the need to pilot these procedures, ensuring they are as simple as possible and reflect a practical evaluation of the functionality intended to support policy goals.
Next, Doug Fridsma and Rich Elmore gave an ONC update. Rich Elmore described the Query Health initiative, as referenced in my previous blog post about sending questions to data (rather than sending data to registries).
The committee endorsed the work and noted that further research will be needed to link patients across multiple databases to avoid double counting individuals in quality measure denominators. The work of Jeff Jonas, as described in my earlier blog post about linking identity.
Doug updated the committee about the S&I Framework initiatives - Transitions of Care, Lab Results, Provider Directories, Data Segmentation (for privacy protection), and electronic submission of medical documentation for Medicare review.
We then discussed a preliminary framework for HITSC 2012 Workplan to ensure the items in the standards trajectory listed above are completed in 2012 as we continue to prepare for meaningful use stage 3.
A great meeting.
We began the meeting by relating our standards trajectory to today's agenda.
Our outstanding standards issues for discussion include
1. Content
Continued discussion of GreenCDA on the wire and overview of Stan Huff's CIMI initiative
Standardizing DICOM image objects for image sharing and investigating other possible approaches (e.g., cloud based JPEG2000 exchange). Consider image transfer standards, image viewing standards, and image reporting standards.
Query Health i.e. I2B2 distributed queries that send questions to data instead of requiring consolidation of data
Simplify the specification for quality measures to enhance consistency of implementation.
The December meeting included an overview of Query Health and Quality measure standards, leaving the discussion of GreenCDA/CIMI and DICOM to our 2012 meetings.
2. Vocabulary
Extend the quality measurement vocabularies to clinical summaries
Lab ordering compendium
The December meeting included a discussion of the lab ordering compendium, leaving the discussion of clinical summary vocabularies to our 2012 meetings.
3. Transport
Specify how the metadata ANPRM be integrated into the health exchange architecture
Additional NwHIN standards development (hearing re Exchange specification complexity, review/oversight of the S&I framework work on Exchange specifications simplification). Further define secure RESTful transport standards.
Accelerate provider directory pilots (Microdata, RESTful query/response that separates the transaction layer from the schema) and rapidly disseminate lessons learned.
The December meeting included an update on the provider directory and certificate components of transport
Our first presentation was an NCVHS update on ACA Section 10109 by Walter Suarez.
The Committee emphasized the need to coordinate NCHVS work and HITSC work given that division between administrative and clinical data is becoming less distinct over time
Our second presentation was an Implementation Workgroup Update by Liz Johnson about testing procedures that support the certification process.
The committee emphasized the need to pilot these procedures, ensuring they are as simple as possible and reflect a practical evaluation of the functionality intended to support policy goals.
Next, Doug Fridsma and Rich Elmore gave an ONC update. Rich Elmore described the Query Health initiative, as referenced in my previous blog post about sending questions to data (rather than sending data to registries).
The committee endorsed the work and noted that further research will be needed to link patients across multiple databases to avoid double counting individuals in quality measure denominators. The work of Jeff Jonas, as described in my earlier blog post about linking identity.
Doug updated the committee about the S&I Framework initiatives - Transitions of Care, Lab Results, Provider Directories, Data Segmentation (for privacy protection), and electronic submission of medical documentation for Medicare review.
We then discussed a preliminary framework for HITSC 2012 Workplan to ensure the items in the standards trajectory listed above are completed in 2012 as we continue to prepare for meaningful use stage 3.
A great meeting.
Labels:
HIT Standards Committee
Monday, December 12, 2011
The Elephant in the Room
Why aren't we talking about pricing failures? The US, has consistently higher prices than any other country. The 2010 report by the International Federation of Health Plans consists of 23 pricing measures and the pattern is the same across each of these measures. And a 2010 investigation of Health Care Cost Trends and Cost Drivers in Massachusetts found that "price variations are correlated to market leverage..."
Before his departure from CMS, Don Berwick was interviewed by the New York Times and took a "parting shot at waste". Berwick listed five elements of waste including overtreatment of patients, failure to coordinate care, administrative complexity, burdensome rules and fraud. Pricing failures didn't make the list. (Many folks have commented and analyzed the five factors including John Halamka's terrific piece on how EHRs can address these 5 factors.)
Then in Berwick's December 7th speech to the IHI National Forum, he adds a sixth element:
Don Berwick defines pricing failures as "the waste that comes as prices migrate far from the actual costs of production plus fair profits." Think about that: "far from the actual costs of production plus fair profits". At a time when total healthcare expenditures consume a huge share of GDP and increasing at rates higher than inflation and wage increases, why haven't pricing failures been on the table? As we struggle to control costs and improve quality, there is intense focus on utilization, regulation and care coordination. Why not also focus on pricing failures?
So why hasn't pricing failures been part of the conversation up to now? Here's how the conversation usually proceeds: Health Affairs November 2011 article, Large Variations In Medicare Payments For Surgery Highlight Savings Potential From Bundled Payment Programs, "found that current Medicare episode payments for certain inpatient procedures varied by 49–130 percent across hospitals sorted into five payment groups. Intentional differences in payments attributable to such factors as geography or illness severity explained much of this variation. But after adjustment for these differences, per episode payments to the highest-cost hospitals were higher than those to the lowest-cost facilities by up to $2,549 for colectomy and $7,759 for back surgery."
Sounds like a clarion call for a focus on pricing failures doesn't it? Actually, no... The authors conclusions only speak to cost efficiency and utilization. "Our study suggests that bundled payments could yield sizable savings for payers, although the effect on individual institutions will vary because hospitals that were relatively expensive for one procedure were often relatively inexpensive for others. More broadly, our data suggest that many hospitals have considerable room to improve their cost efficiency for inpatient surgery and should look for patterns of excess utilization, particularly among surgical specialties, other inpatient specialist consultations, and various types of postdischarge care."
So is it time to broaden the conversation to include pricing failures? At least one health system has realized that "the jig is up". Perhaps it's time to peel the onion a bit... And take a serious look at pricing failures which deviate "far from the actual costs of production plus fair profits".
_______________________________
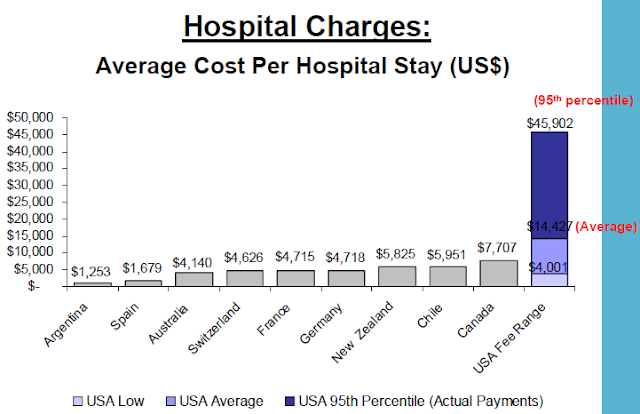
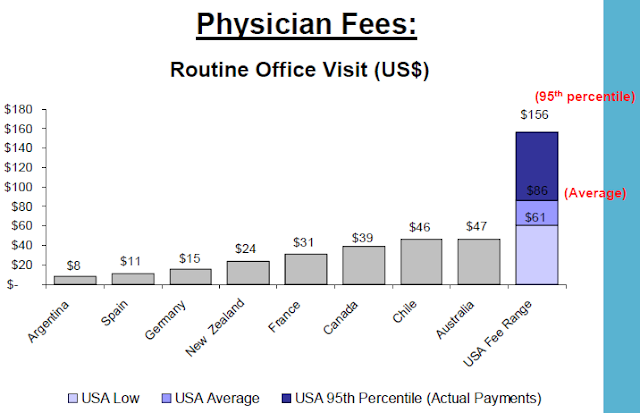
Background tables from the IFHP report:
Before his departure from CMS, Don Berwick was interviewed by the New York Times and took a "parting shot at waste". Berwick listed five elements of waste including overtreatment of patients, failure to coordinate care, administrative complexity, burdensome rules and fraud. Pricing failures didn't make the list. (Many folks have commented and analyzed the five factors including John Halamka's terrific piece on how EHRs can address these 5 factors.)
Then in Berwick's December 7th speech to the IHI National Forum, he adds a sixth element:
- "Overtreatment – the waste that comes from subjecting people to care that cannot possibly help them – care rooted in outmoded habits, supply-driven behaviors, and ignoring science.
- Failures of Coordination- the waste that comes when people – especially people with chronic illness – fall through the slats. They get lost, forgotten, confused. The result: complications, decays in functional status, hospital readmissions, and dependency.
- Failures of Reliability – the waste that comes with poor execution of what we know to do. The result: safety hazards and worse outcomes.
- Administrative Complexity – the waste that comes when we create our own rules that force people to do things that make no sense – that converts valuable nursing time into meaningless charting rituals or limited physician time into nonsensical and complex billing procedures.
- Pricing Failures – the waste that comes as prices migrate far from the actual costs of production plus fair profits.
- Fraud and Abuse – the waste that comes as thieves steal what is not theirs, and also from the blunt procedures of inspection and regulation that infect everyone because of the misbehaviors of a very few. We have estimated how big this waste is – from both the perspective of the Federal payers – Medicare and Medicaid – and for all payers."
Don Berwick defines pricing failures as "the waste that comes as prices migrate far from the actual costs of production plus fair profits." Think about that: "far from the actual costs of production plus fair profits". At a time when total healthcare expenditures consume a huge share of GDP and increasing at rates higher than inflation and wage increases, why haven't pricing failures been on the table? As we struggle to control costs and improve quality, there is intense focus on utilization, regulation and care coordination. Why not also focus on pricing failures?
So why hasn't pricing failures been part of the conversation up to now? Here's how the conversation usually proceeds: Health Affairs November 2011 article, Large Variations In Medicare Payments For Surgery Highlight Savings Potential From Bundled Payment Programs, "found that current Medicare episode payments for certain inpatient procedures varied by 49–130 percent across hospitals sorted into five payment groups. Intentional differences in payments attributable to such factors as geography or illness severity explained much of this variation. But after adjustment for these differences, per episode payments to the highest-cost hospitals were higher than those to the lowest-cost facilities by up to $2,549 for colectomy and $7,759 for back surgery."
Sounds like a clarion call for a focus on pricing failures doesn't it? Actually, no... The authors conclusions only speak to cost efficiency and utilization. "Our study suggests that bundled payments could yield sizable savings for payers, although the effect on individual institutions will vary because hospitals that were relatively expensive for one procedure were often relatively inexpensive for others. More broadly, our data suggest that many hospitals have considerable room to improve their cost efficiency for inpatient surgery and should look for patterns of excess utilization, particularly among surgical specialties, other inpatient specialist consultations, and various types of postdischarge care."
So is it time to broaden the conversation to include pricing failures? At least one health system has realized that "the jig is up". Perhaps it's time to peel the onion a bit... And take a serious look at pricing failures which deviate "far from the actual costs of production plus fair profits".
_______________________________
Background tables from the IFHP report:
Monday, November 21, 2011
The November HIT Standards Committee
by John Halamka, Life as a Healthcare CIO, November 16, 2011
Today, the HIT Standards Committee shifted gears from the Summer Camp work on Meaningful Use Stage 2 and began new interoperability efforts.
We began the meeting with a presentation by Liz Johnson and Judy Murphy about the Implementation Workgroup's recommendations to improve the certification and testing process. These 15 items incorporate the Stage 1 experience gathered from numerous hospitals and eligible professionals. If ONC and NIST can implement this plan, many stakeholders will benefit. The Committee approved these recommendations without revision.
Next, we focused on content, vocabulary and transport standards.
In my October HIT Standards Committee blog post, I noted that HITSC should work on the following projects:
Content
*Continued refinement of the Consolidated CDA implementation guides and tools to enhance semantic interoperability including consistent use of business names in "Green" over-the-wire standards.
*Simplifying the specification for quality measures to enhance consistency of implementation.
*Standardizing DICOM image objects for image sharing and investigating other possible approaches. We'll review image transfer standards, image viewing standards, and image reporting standards.
*Query Health - distributed queries that send questions to data instead of requiring consolidation of the data
Vocabulary
*Extending the quality measurement vocabularies to clinical summaries
*Finalizing a standardized lab ordering compendium
Transport
*Specifying how the metadata ANPRM be integrated into health exchange architectures
*Supporting additional NwHIN standards development (hearings about Exchange specification complexity, review/oversight of the S&I Framework projects on simplification of Exchange specifications). Further defining secure RESTful transport standards.
*Accelerating provider directory pilots (Microdata, RESTful query/response that separates the transaction layer from the schema) and rapidly disseminating lessons learned.
The November Committee agenda included a discussion of Consolidated CDA, Quality Measures, and NwHIN Implementation Guides.
Doug Fridsma began with a discussion of the Consolidated CDAwork and the tools which support it.
The Committee had a remarkable dialog with more passion and unanimity than at any recent discussion. We concluded:
*Simple XML that is easily implemented will accelerate adoption
*That simple XML should be backed by a robust information model. However, implementers should not need expert knowledge of that model. The information model can serve as a reference for SDOs to guide their work
*Detailed Clinical Models, as exemplified by Stan Huff's Clinical Information Modeling Initiative (CIMI) hold great promise. Stan has assembled an international consensus group including those who work on
-Archetype Object Model/ADL 1.5 openEHR
-CEN/ISO 13606 AOM ADL 1.4
-UML 2.x + OCL + healthcare extensions
-OWL 2.0 + healthcare profiles and extensions
-MIF 2 + tools HL7 RIM – static model designer
Their work may be much more intuitive than today's HL7 RIM as the basis for future clinical exchange standards.
*Rather than debate whether Consolidated CDA OR GreenCDA(simplified XML tagging) should be the over the wire format, the Committee noted that "OR" really implies "AND" for vendors and increases implementation burden. The Committee endorsed moving forward with GreenCDA as the single over the wire format.
*We should move forward now with this work, realizing that it will take 9-12 months and likely will not be included in Meaningful Use Stage 2, but it is the right thing to do.
Thus, the future Transfer of Care Summary will be assembled from a simple set of clinically relevant GreenCDA templates, based on CIMI models, as needed to support various use cases. There will be no optionality - just a single way to express medical concepts in specific templates.
To support this approach, we'll need great modeling tools. David Carlson and John Timm presented the applications developed to support the VA's Model Driven Health Tools initiative. This software turns clinical models into XML and conformance testing tools. The committee was very impressed.
Next, Avinash Shanbhag presented the ONC work on Quality Measures that seeks to ensure quality numerators and denominators are expressed in terms of existing EHR data elements captured as part of standard patient care workflows.
Avinash also presented an update on transport efforts, which include easy to use, well documented implementation guides for SMTP/SMIME and SOAP. The work is highly modular and does not require that the full suite of NwHIN Exchange specifications be implemented for SOAP exchanges.
As part of the ongoing efforts to improve NwHIN Exchange, the HIT Standards Committee is seeking input from NwHIN implementers per this blog post.
Finally, Wil Yu updated the committee on the SHARP and other innovation programs.
There will be a great body of challenging work to do in 2012. What's needed after that? The next 5 years will include many new regulations as healthcare reform is rolled out. It's clear that the Standards Committee will have many topics to discuss.
Today, the HIT Standards Committee shifted gears from the Summer Camp work on Meaningful Use Stage 2 and began new interoperability efforts.
We began the meeting with a presentation by Liz Johnson and Judy Murphy about the Implementation Workgroup's recommendations to improve the certification and testing process. These 15 items incorporate the Stage 1 experience gathered from numerous hospitals and eligible professionals. If ONC and NIST can implement this plan, many stakeholders will benefit. The Committee approved these recommendations without revision.
Next, we focused on content, vocabulary and transport standards.
In my October HIT Standards Committee blog post, I noted that HITSC should work on the following projects:
Content
*Continued refinement of the Consolidated CDA implementation guides and tools to enhance semantic interoperability including consistent use of business names in "Green" over-the-wire standards.
*Simplifying the specification for quality measures to enhance consistency of implementation.
*Standardizing DICOM image objects for image sharing and investigating other possible approaches. We'll review image transfer standards, image viewing standards, and image reporting standards.
*Query Health - distributed queries that send questions to data instead of requiring consolidation of the data
Vocabulary
*Extending the quality measurement vocabularies to clinical summaries
*Finalizing a standardized lab ordering compendium
Transport
*Specifying how the metadata ANPRM be integrated into health exchange architectures
*Supporting additional NwHIN standards development (hearings about Exchange specification complexity, review/oversight of the S&I Framework projects on simplification of Exchange specifications). Further defining secure RESTful transport standards.
*Accelerating provider directory pilots (Microdata, RESTful query/response that separates the transaction layer from the schema) and rapidly disseminating lessons learned.
The November Committee agenda included a discussion of Consolidated CDA, Quality Measures, and NwHIN Implementation Guides.
Doug Fridsma began with a discussion of the Consolidated CDAwork and the tools which support it.
The Committee had a remarkable dialog with more passion and unanimity than at any recent discussion. We concluded:
*Simple XML that is easily implemented will accelerate adoption
*That simple XML should be backed by a robust information model. However, implementers should not need expert knowledge of that model. The information model can serve as a reference for SDOs to guide their work
*Detailed Clinical Models, as exemplified by Stan Huff's Clinical Information Modeling Initiative (CIMI) hold great promise. Stan has assembled an international consensus group including those who work on
-Archetype Object Model/ADL 1.5 openEHR
-CEN/ISO 13606 AOM ADL 1.4
-UML 2.x + OCL + healthcare extensions
-OWL 2.0 + healthcare profiles and extensions
-MIF 2 + tools HL7 RIM – static model designer
Their work may be much more intuitive than today's HL7 RIM as the basis for future clinical exchange standards.
*Rather than debate whether Consolidated CDA OR GreenCDA(simplified XML tagging) should be the over the wire format, the Committee noted that "OR" really implies "AND" for vendors and increases implementation burden. The Committee endorsed moving forward with GreenCDA as the single over the wire format.
*We should move forward now with this work, realizing that it will take 9-12 months and likely will not be included in Meaningful Use Stage 2, but it is the right thing to do.
Thus, the future Transfer of Care Summary will be assembled from a simple set of clinically relevant GreenCDA templates, based on CIMI models, as needed to support various use cases. There will be no optionality - just a single way to express medical concepts in specific templates.
To support this approach, we'll need great modeling tools. David Carlson and John Timm presented the applications developed to support the VA's Model Driven Health Tools initiative. This software turns clinical models into XML and conformance testing tools. The committee was very impressed.
Next, Avinash Shanbhag presented the ONC work on Quality Measures that seeks to ensure quality numerators and denominators are expressed in terms of existing EHR data elements captured as part of standard patient care workflows.
Avinash also presented an update on transport efforts, which include easy to use, well documented implementation guides for SMTP/SMIME and SOAP. The work is highly modular and does not require that the full suite of NwHIN Exchange specifications be implemented for SOAP exchanges.
As part of the ongoing efforts to improve NwHIN Exchange, the HIT Standards Committee is seeking input from NwHIN implementers per this blog post.
Finally, Wil Yu updated the committee on the SHARP and other innovation programs.
There will be a great body of challenging work to do in 2012. What's needed after that? The next 5 years will include many new regulations as healthcare reform is rolled out. It's clear that the Standards Committee will have many topics to discuss.
Labels:
HIT Standards Committee,
John Halamka
Monday, November 14, 2011
The Elephant in the Room: The Prequel
At the October HIT Policy Committee, Charles Kennedy described his work with health systems establishing accountable care models. His clients "have actual health plan products that are private labeled products with the delivery systems' name on it that they’re selling."
Kenedy talked with the COO of one health system that was particularly high cost. Kennedy asked the COO: "Why on earth would you want to form an ACO? You’re a monopoly. You’re making tons of money. You can keep doing this for some period of time."
The COO replied “Look I understand that the jig is up.”
The COO went on to say "I know how to take $60 out per member per month. $60 - - out of my cost structure. I know exactly how to do it. I never had a motivation to do it before - - until health care reform happened." Kennedy explained that the COO has now "taken those costs out of his delivery system and because he has a product in the marketplace he gets to reap those efficiencies. The second thing he said was that 'I never really had a use for health IT until I began to take costs out of my infrastructure'."
Kenedy talked with the COO of one health system that was particularly high cost. Kennedy asked the COO: "Why on earth would you want to form an ACO? You’re a monopoly. You’re making tons of money. You can keep doing this for some period of time."
The COO replied “Look I understand that the jig is up.”
The COO went on to say "I know how to take $60 out per member per month. $60 - - out of my cost structure. I know exactly how to do it. I never had a motivation to do it before - - until health care reform happened." Kennedy explained that the COO has now "taken those costs out of his delivery system and because he has a product in the marketplace he gets to reap those efficiencies. The second thing he said was that 'I never really had a use for health IT until I began to take costs out of my infrastructure'."
Subscribe to:
Posts (Atom)